 
Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron. Niels Bohr’s atomic model was created based on previous research by Rutherford, Rutherford’s gold foil experiment, and. It was a postulation of Bohr that the electrons rotated in a circular orbit around the nucleus of the atom. The intensity of spectral lies could not be clarified by Bohr. Orbits were presumed to be circular but Sommerfield further stated that these orbits are elliptical. Bohr ’s model and the quantum atomic model changed the way we see the atom ’ s nature and how electrons interact within it. Rutherford did not explain the relation of the orbital. Successive atomic models, such as those proposed by Thomson and Rutherford, changed the way we think about the atom ’s charge, as they included electrical charges and described how these were distributed in the atom. 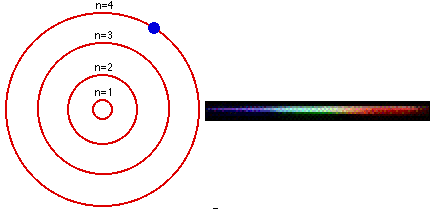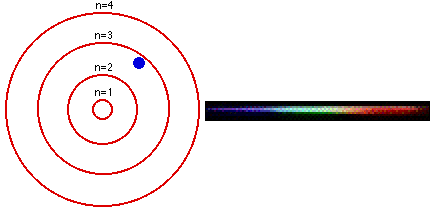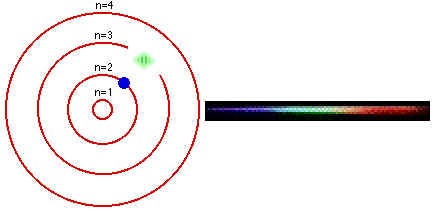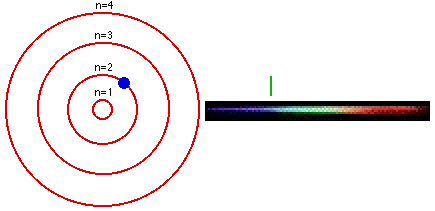
Bohr’s atomic theory was based on the Hydrogen line spectrum, while Rutherford’s atomic theory was on Gold foil. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. The Niels Bohr Atomic Model theory is a model that was introduced by Niels Bohr in 1913 to describe the atom. Bohr’s theory of the atomic model was quite effective in explaining the stability of the atom and the line spectrum of a hydrogen atom. Postulates were given in the year Niels Bohr in 1922, Ernest Rutherford in 1913. 
In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. Atoms are the basic units of chemical elements and were once believed to be the smallest indivisible structures of matter. This atomic model is the modification of Rutherford’s atomic model (the nucleus is positively charged and is surrounded by electrons (negatively charged particles). Where \(n_1\) and \(n_2\) are positive integers, \(n_2 > n_1\), and \( \Re \) the Rydberg constant, has a value of 1.09737 × 10 7 m −1. Bohr’s model consists of a small nucleus (positively charged) surrounded by negative electrons moving around the nucleus in orbits. Bohr's ill - defined ideas on atomic motion and radiation processes, other physicists tended to regard his theory as clever bricolage, employing tools. In 1913, the physicist Niels Bohr introduced a model of the atom that contributed a greater understanding to its structure and quantum mechanics. In 1913, Danish physicist Neil Bohr proposed the Bohr atomic model based on Planck’s quantum theory of radiation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
